 
Sulfur has six valence electrons in its outer shell. Total number of valence electrons for SF 2 – Valence electrons of Sulphur + Valence electrons of Fluorine So we will first find out the total valence electrons for Sulphur Difluoride.  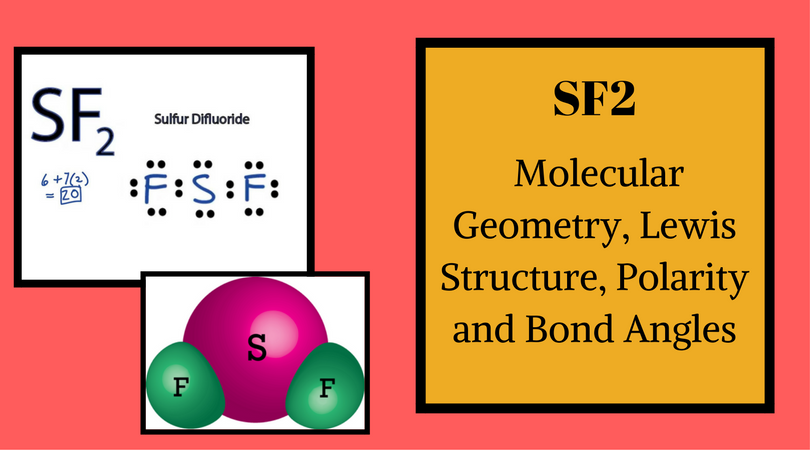
For drawing the Lewis structure for any molecule, we first need to know the total number of valence electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
